|
2,4-dioxa-3-thia-5-alpha-androstane, a forgotten eccentric anabolic from the seventies
Between 2004 and 2016, during the heyday of designer steroids, the rogue chemists from the supplement industry decided to ignore the anabolic steroids designed by Galal Zanati and Manfred Wolff, two pharmacologists from the University of California at San Francisco. Their steroids were extremely interesting though. Zanati and Wolff made anabolics that you would think could not do anything at all.
Oxygen instead of carbon
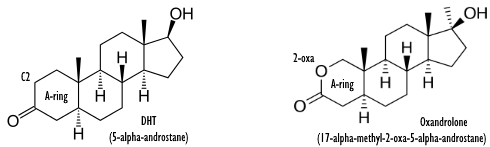
In the 1960s, Searle introduced oxandrolone: an oral anabolic steroid with remarkably few side effects. You can describe oxandrolone as an orally available analogue of DHT, with a skeleton whose second carbon atom has been replaced by an oxygen atom.
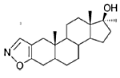
What happens if you replace even more carbon atoms in the A-ring of the androstane skeleton with oxygen atoms? Zanati and Wolff found that question interesting enough to make a few of those things and test them on laboratory rats.
Hersberger assay
This testing was done using a method developed by biologists from the University of Wisconsin in the early 1950s, the Hershberger assay. [Proc Soc Exp Biol Med. 1953;83(1):175-80.]
The method involves treating a group of castrated rats with testosterone or an analogue thereof for a period of a few days, and another group of rats with the anabolicum to be tested. You then compare the effect of both drugs on the weight of prostate and the weight of the sphincter or levator ani.
The effect on the levator ani gives an idea of the desired anabolic effect of an anabolic steroid. The effect on the prostate gives an idea of the unwanted androgenic side effects. Between 1935 and 1980, chemists tried to make anabolics with more anabolic effect than testosterone - but with fewer androgenic effects.
Results
Zanati and Wolff made the 3 variants that you see below, and injected them into their test animals. Compound 4 did not do much, but compound 3 and especially compound 5 were active.


The anabolic effect of compound 5 is probably about the same as that of testosterone, the undesirable androgenic side effects are half that of testosterone.
The researchers used acetates. Perhaps their results would have been impressive if they had used other esters, such as enantates or decanoates.
It is unlikely that 2,4-dioxa-3-thia-5-alpha-androstane ever comes on the market. The thing is probably way too expensive to produce. But hey - it was a nice idea.
Source:
J Med Chem. 1971;14(10):961-2.
More:
What is steroid 36644-Ba? 01.01.2020
What are dehydroandrostenol and dehydroandrosterone? 16.03.2019
What kind of anabolic steroid is methyldiazirinol? 22.10.2018
Archives:
Designer Steroids
Anabolic Steroids
|