|
One dose of myostatin inhibitor bimagrumab results in permanent muscle mass increase
People with type-2 diabetes can build up a couple of kilos of muscle mass and lose a couple of kilos of fat, without training or changing their diet, by taking a one-time dose of the myostatin inhibitor bimagrumab. A study by researchers at Novartis has shown this to be the case.
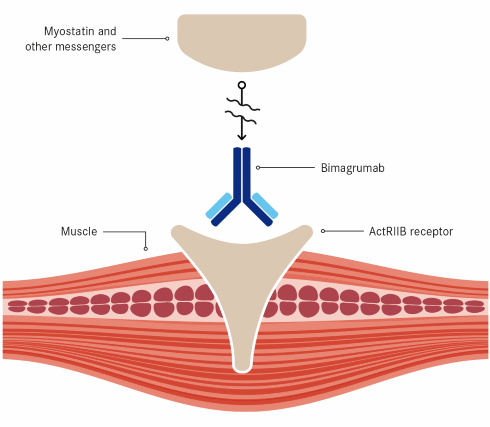
Bimagrumab
Bimagrumab is also called BYM338. It is an antibody, developed by Novartis, which attaches itself to the activin type II receptor-A and activin type II receptor-B. This prevents muscle-growth inhibiting proteins such as myostatin, GDF-11 and activin from working, and as a result muscles grow bigger and stronger.
The pharmaceutical industry has been experimenting for years with myostatin inhibitors, and some of them are already marketed as 'research chemicals'. These grey market inhibitors are not particularly effective, however. Apart from a few substances, it is mostly the pharmaceutical industry rejects that athletes manage to get their hands on in this way.
Going by the promising human studies published, bimagrub is certainly not a reject. [J Cachexia Sarcopenia Muscle. 2017 Sep 14. doi: 10.1002/jcsm.12205. [Epub ahead of print].] [J Am Geriatr Soc. 2017 Jun 27. doi: 10.1111/jgs.14927. [Epub ahead of print].] [Neurology. 2014 Dec 9;83(24):2239-46.]
Study
The researchers gave 10 overweight people with type-2 diabetes 30 mg bimagrumab per kg bodyweight intravenously. A similar control group was given a placebo.
Results
In week 14 the lean body mass of the participants who had been given bimagrumab had increased by 5 percent since week 0, and their body fat had decreased by 10 percent.
The effects were still visible six months after the bimagrumab had been administered.

The insulin sensitivity and glucose balance of the participants who had received bimagrumab improved.
Side effects
"Acne, muscle spasms and myalgia were reported in 30% of bimagrumab-treated participants and in none of the placebo-treated participants; there were no changes in creatine kinase levels," wrote the researchers. "Diarrhoea, muscle weakness and musculoskeletal stiffness were noted in 10% of the bimagrumab group and in none of the placebo group."
"All events were described as minor and transient and resolved spontaneously by the end of the trial."
"Overall, bimagrumab was safe and well tolerated".
Conclusion
"Bimagrumab has potential to be developed for metabolic complications of obesity such as insulin resistance, although this was only an exploratory study," the researchers concluded.
"A trial with bimagrumab in addition to caloric restriction in obese patients with type-2 diabetes is ongoing, with the aim of evaluating the translatability of these pilot findings on body composition, body weight and metabolic variability." [clinicaltrials.gov]
"It is possible that bimagrumab could be a novel insulin-sensitizing therapeutic agent for type-2 diabetes through its meaningful effect on body composition. Unlike the available therapeutics for type-2 diabetes, which tend to increase weight and fat mass, bimagrumab could reverse important features of the underlying pathophysiology by having a positive effect on body composition in people with type-2 diabetes, and improve insulin sensitivity."
Source:
Diabetes Obes Metab. 2017 Jun 23. doi: 10.1111/dom.13042. [Epub ahead of print].
More:
Decorin - myostatin inhibitor and follistatin booster - is key factor in muscle growth 19.11.2014
Animal study: a year of growth after just one anti-myostatin gene treatment 16.06.2013
Research on myostatin inhibitor ACE-031 stopped - but the stuff does work 14.06.2013
Archives:
Blocking Myostatin
|